|
But considering how long ago this was, it was a pretty good start. While he was on the right track, his ideas on matter weren’t rigorously scientific for example, he imagined bitter atoms to be sharp and pointy and sweet atoms to be round and smooth. He extrapolated this process, reasoning that he would eventually get to a particle that he could no longer cut but that would have the same properties as the stone. If he broke those in two, the four pieces would also have the same properties. If he broke it in two, each half would have the same properties. Instead, he conducted more of a thought experiment. A few things to note about his work: Democritus was a philosopher, not a scientist, and he didn’t conduct physical experiments in the way that we do today. He called them atomos, meaning “indivisible,” which is where we get our modern word atom. He reasoned that all materials are made up of small, uncuttable units. We’ll begin our story 2,400 years ago with the Greek philosopher Democritus. So, try to be careful and mentally mark which parts of each theory were right and which were wrong!Īlright, with that, let’s dive into the exciting history of the atomic model! Democritus Sometimes just by hearing something, even if you know it’s wrong, it will stick in your brain as true. 
The important thing to remember is that you should make an effort to mentally label which theories are wrong. And, taking this historical look at the development of the atomic model is a great way to learn about the scientific method. So why not just ignore the older, incorrect models? Because it is important to note that these early scientists actually got some things right and understanding the things that they got wrong can also be instructive. This is all part of the scientific process and it makes science exciting. As time went on, new scientists examined those findings, found inconsistencies, ran new experiments, and came to new conclusions. Then they made new hypotheses based on those conclusions but weren’t really able to test them or didn’t even realize they needed to be tested. They ran experiments and came to conclusions. 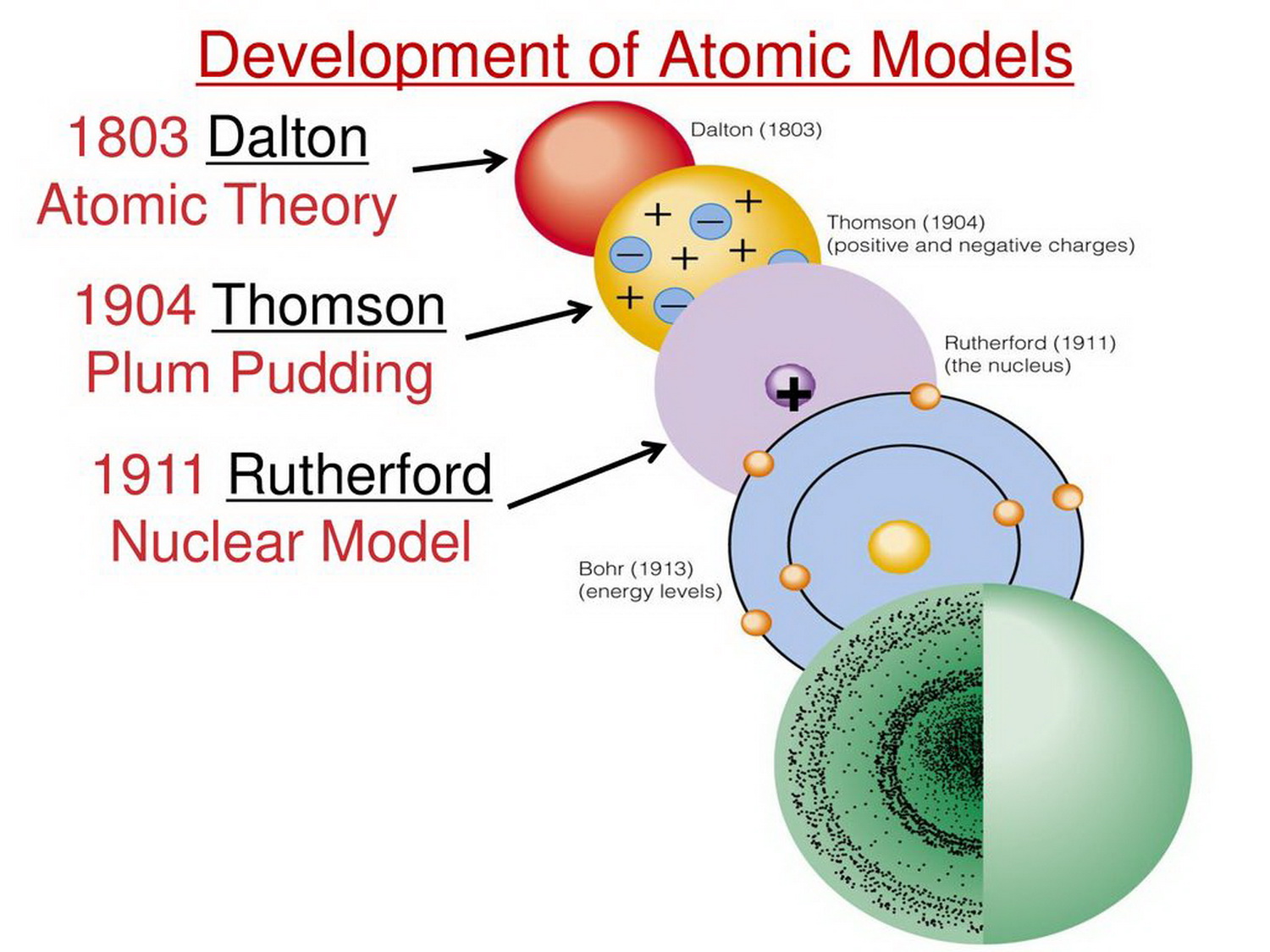
While these were amazing scientists, they did make mistakes. What if you didn’t have a smartphone or computer or any modern instrumentation to examine matter? How would you even begin going about proving the existence of the atom and describing its subatomic structure? Pretty unimaginable, right? These were amazing scientists back in the day.Īnd second. As we go through the history, try to imagine what it was like to live in the different time periods. Let’s get started!īefore we begin, we need to consider two things.įirst. Then, we’ll launch forward in time to the 1800s and learn how the modern description of the atom was derived. We’ll start way back in 400 BC when the Greek philosophers postulated that matter was made up of small, discrete units. Hi, and welcome to this review of atomic models! Today, we’re going to be discussing the atomic model and the experiments that led to its development.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
